CARDIOVASCULAR & METABOLIC
Diuretic-resistant congestion – ‘The road less travelled’ in novel treatments for chronic heart failure
Ian Crosbie, CEO at Sequana Medical, explores treatments for chronic heart failure and the challenges facing their development
Congestion is a key challenge in heart failure
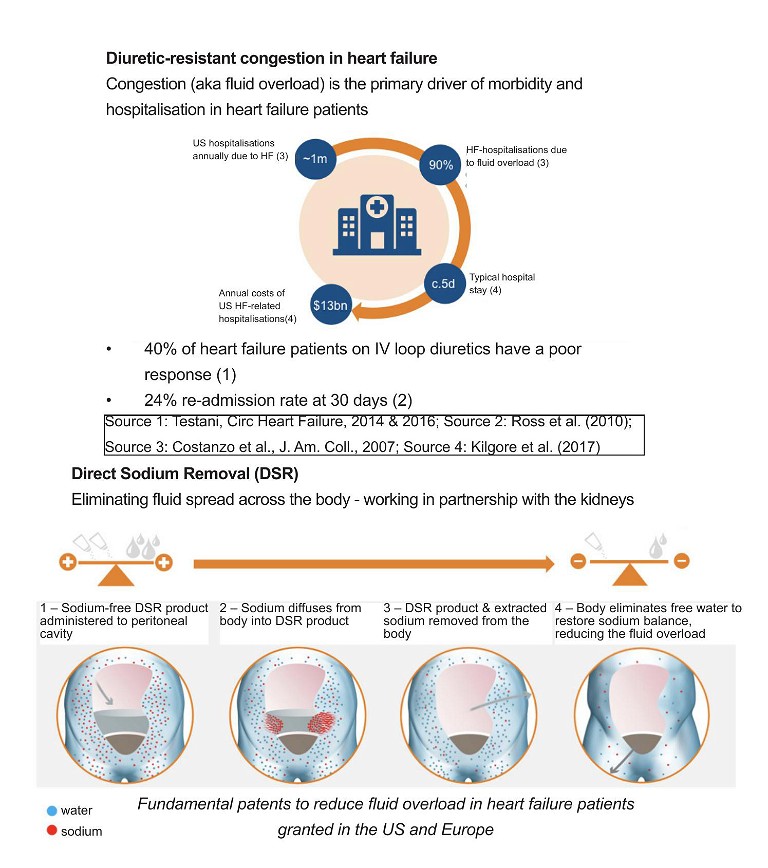
The problem of heart failure (HF) is well recognised, with The American Heart Association estimating that 6.5 million adults in the US are affected by HF, and that number is expected to rise to 8 million adults by 2030. Overall chronic heart failure (CHF) death rates remain significantly high, with up to half of patients dying within five years of a HF diagnosis. Although there have been tremendous advances in both drug and device approaches in recent years, they have generally focused on improving the function of the failing heart whilst failing to address the key issue of congestion. While HF is generally regarded as the inability of the heart to pump sufficient blood, on a population level, congestion (aka, hypervolemia/volume overload) is the primary driver of morbidity and hospitalisation. Symptoms and signs of volume overload directly result from the mechanical consequences of fluid (water) retention. However, the primary pathophysiologic driver of water accumulation is actually sodium retention, with fluid largely passively following the retained salt.
Diuretics – limitations and resistance
Diuretics and, in particular, loop diuretics (LD) – drugs that force the kidneys to remove excessive salt/water from the body – are the standard of care for this patient group, but there is well-described toxicity and resistance is a major problem.
Notably, some degree of diuretic resistance is essentially the rule in HF, with the average diuretic response being approximately 10% of the normal response to LD. Once patients become significantly diuretic-resistant or are at risk of severe renal damage, there are limited clinical alternatives to combat congestion, and the majority of these are of limited effectiveness.
It is well documented in both animal and human studies that there is adverse structural remodelling of the kidney that occurs with chronic exposure to LD.
Furthermore, the escalating doses required to maintain fluid balance are acknowledged as a double-edged sword (i.e., ‘save the heart or save the kidneys’). In addition, LD strongly activates the neurohormonal system, which not only increases the retention of sodium (and thereby fluid overload) but also acts contrary to the mainstays of HF therapy, such as ARBs, MRAs, betablockers and ACE-inhibitors, which seek to block the neurohormonal system.
HF is the leading cause of hospitalisations in patients over 65 in the US and, of these 1 million hospitalisations for HF patients each year in the US, 90% are due to fluid overload. The ADHERE registry highlights that ~50% of patients are discharged with no clinically relevant loss of weight and one in four acutely decompensated HF patients are readmitted within 30 days of discharge. There are an estimated 400,000 HF patients in the US and Europe that are chronically decompensated or congested (the ‘frequent flyers’) and they have few clinical options due to their poor response/inability to tolerate diuretic therapy. These patients have repeated hospitalisations per year, as well as poor clinical outcomes. There is no apparent new drug nor any successor to LD in development for HF patients, and the cost of HF hospitalisations in the US is over $14.5 billion annually.
Novel approach to address this key driver of heart failure
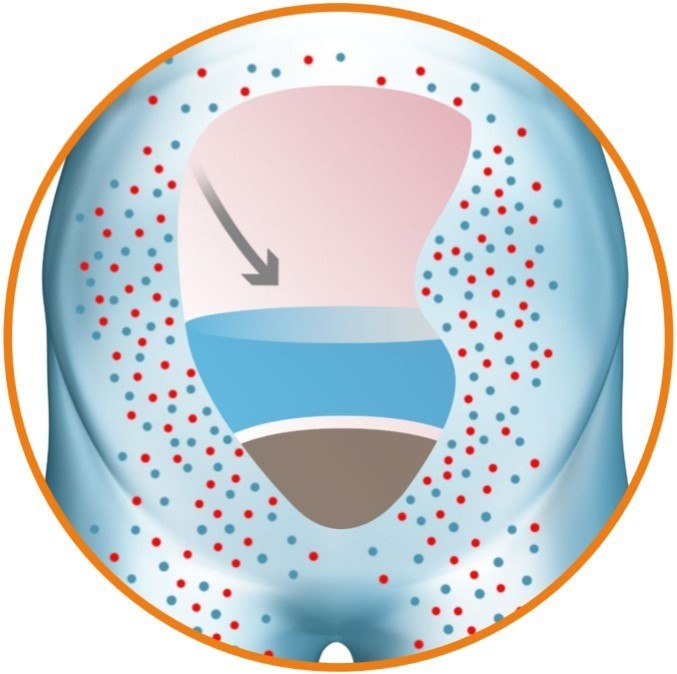
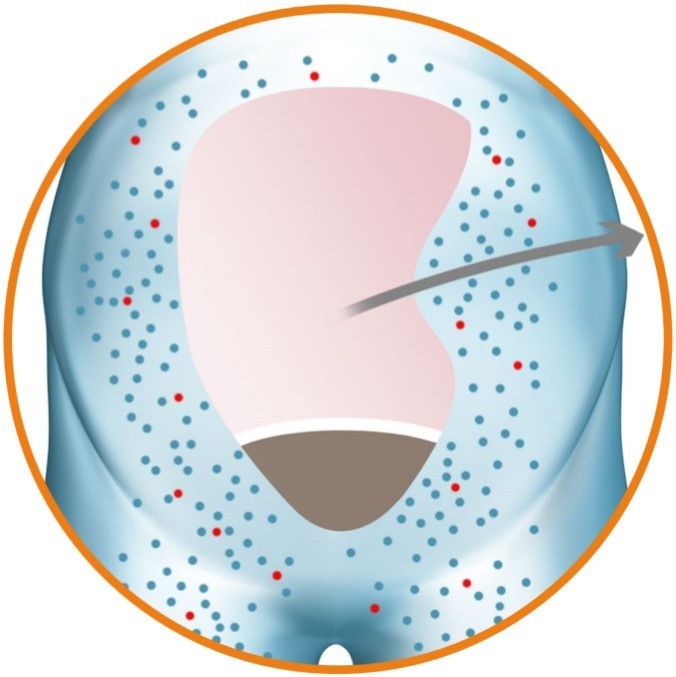
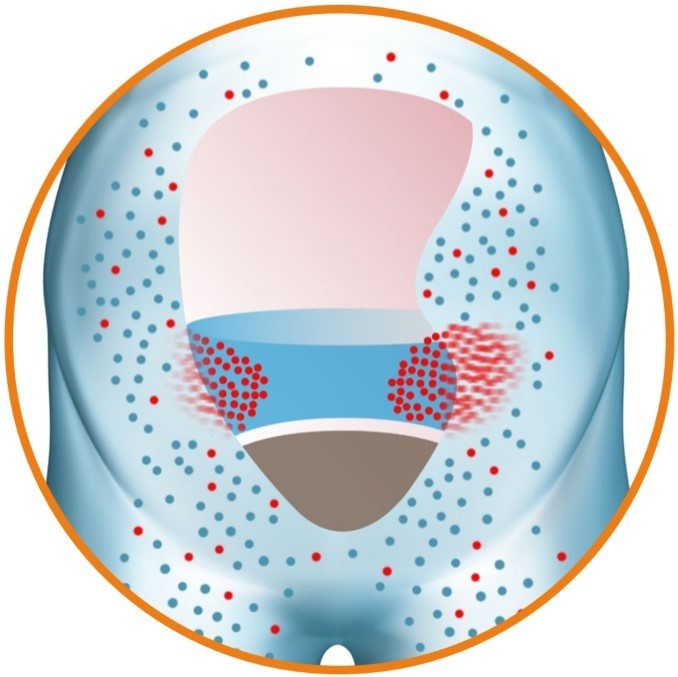
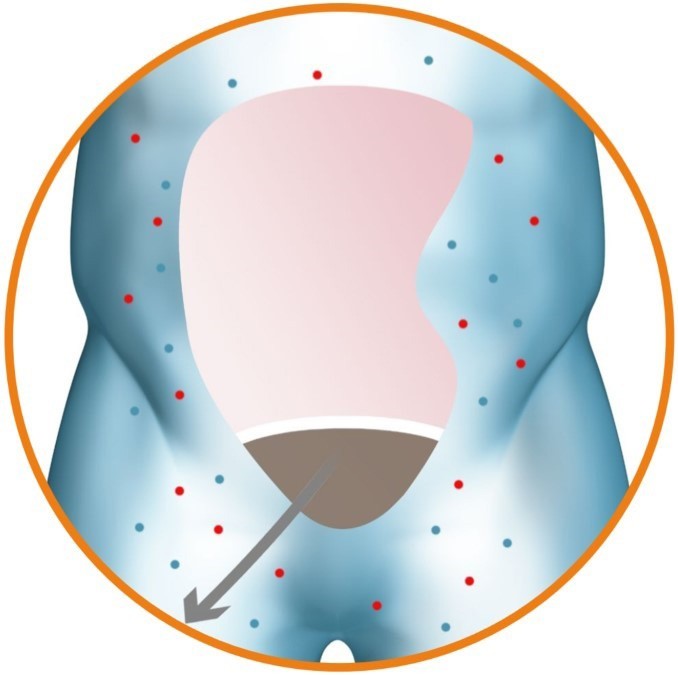
Direct Sodium Removal (DSR), a drug therapy currently in development by Sequana Medical, has the potential to improve the clinical outcomes for this large and growing body of HF patients. DSR therapy works in partnership with the kidneys to safely, rapidly and effectively remove the fluid overload, improve cardio-renal status and restore the ability of the kidney to manage the body’s fluid balance naturally. DSR therapy removes excess sodium from the body, prompting the body to eliminate excess fluid. Removing the need for LD for the duration of DSR therapy allows the cardiorenal system to ‘reset’, restoring the natural ability to manage fluid and sodium for an extended period.
The procedure involves administration of the sodium-free DSR solution into the peritoneal cavity and allowing it to dwell for a pre-defined period of time. During this period, sodium diffuses from the body down a steep gradient into the DSR solution with the circulation keeping the effective blood sodium concentration high. The DSR solution and the extracted sodium are then removed from the body, resulting in a significant elimination of sodium. The body responds to restore the concentration of sodium in the body by eliminating the excess fluid via urination and osmotic ultrafiltration, the movement of water and sodium from the bloodstream to the peritoneal cavity, resulting in reduced fluid overload. No clinically relevant electrolyte imbalances have been observed during clinical studies.
With an estimated 400,000 HF patients in the US and Europe suffering every day from diuretic-resistant fluid overload, there is a desperate need for disease-modifying therapies to address this forgotten driver of heart failure, improve clinical outcomes, transform patient quality of life and reduce the huge costs for healthcare systems.

Ian Crosbie has served as Chief Executive Officer of Sequana Medical since 2016 and has been a member of the Board of Directors since 2019. Ian has over 25 years of experience in the healthcare sector, both in-house at medical device and pharmaceutical companies, and as an investment banker at leading global firms. He has extensive expertise in implantable medical devices and a strong track record in capital markets, licensing and strategic transactions.